- 1. Zitzmann NU, Berglundh T. Definition and prevalence of peri-implant diseases. J Clin Perio. 2008;35:286–291.
- 2. Lazzara RJ†, Porter SS†. Platform Switching: A new concept in implant dentistry for controlling post restorative crestal bone levels. Int J Periodontics Restorative Dent. 2006;26:9-17.
- 3. Fransson C, Lekholm U, Jemt T, Berglundh T. Prevalence of subjects with progressive bone loss at implants. Clinical Oral Implants Research. 2005;16:440–446.
- 4. Suttin Z††, Towse R††. Effect of abutment screw design on implant system seal performance. Presented at the European Association for Osseointegration, 20th Annual Scientific Meeting; October 2012; Copenhagen, Denmark.
- 5. Byrne D, Jacobs S, O’Connell B, Houston F, Claffey N. Preloads generated with repeated tightening in three types of screws used in dental implant assemblies. J. Prosthodont. 2006 May–Jun;15(3):164-171.
- † These clinicians have or had financial relationships with Zimmer Biomet Dental resulting from speaking engagements, consulting engagements and other retained services.
- †† These authors conducted their research while employed at Biomet 3i, LLC.
- * Bench test results do not always necessarily reflect human clinical experience.
DIEM® 2 (Certain® & External Hex Connections)
Immediate Full-Arch Rehabilitation
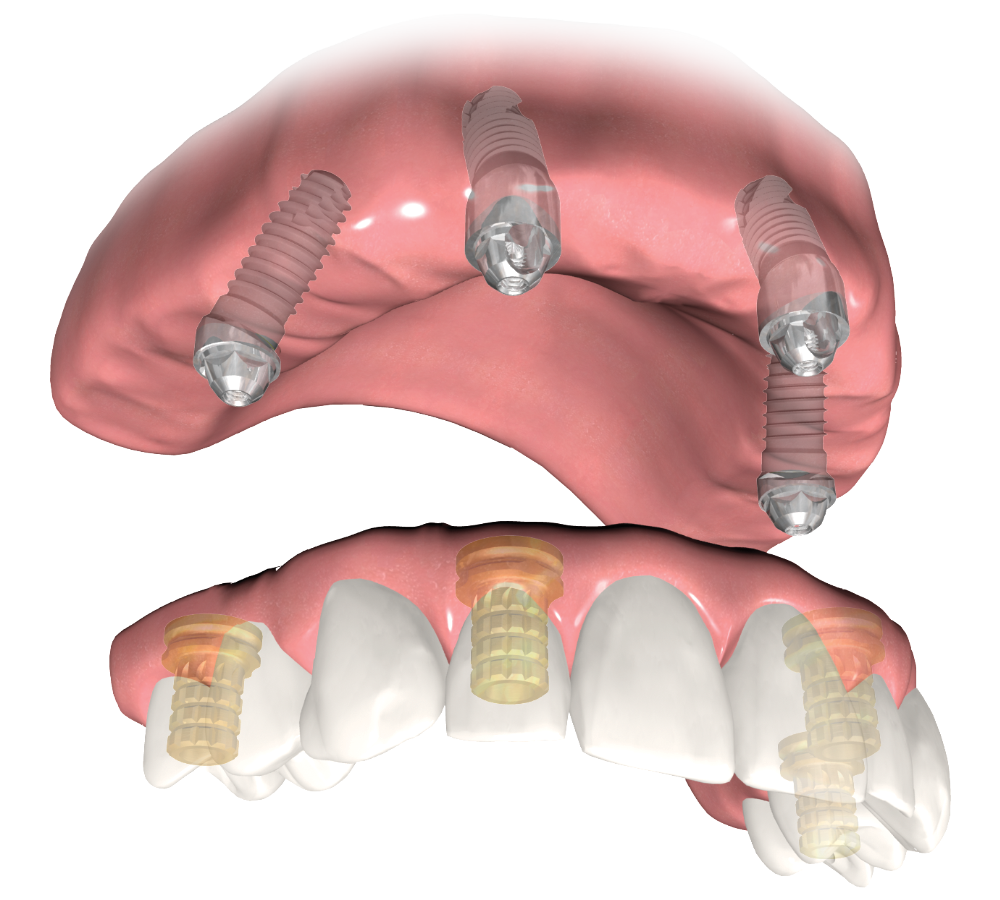
The DIEM 2 Protocol is designed to help provide long-lasting aesthetic results through comprehensive dental implant therapy for edentulous patients, partially edentulous patients and those with failing dentition.
- Edentulism and failing dentition continue to be major health challenges affecting millions of people worldwide.
- Many patients seek full-arch rehabilitation to quickly regain their confidence and quality of life. You can help these patients by offering them DIEM 2, the ZimVie Dental solution for immediate full-arch restoration for one or both arches.
Clinical Challenges
Implant-Abutment Seal Strength with the Gold-Tite Screw
- Peri-implant Mucositis - The prevalence of peri-mucositis has been reported as high as 80% of all dental patients.1
- Peri-implantitis - The prevalence of implants experiencing peri-implantitis has been reported in excess of 12%.2,3
- Crestal Bone Loss - Average implant crestal bone remodeling can exceed 1.5 mm following the first year of function, leading to compromised aesthetics.3
Features & Benefits
Benefits for Clinicians
- Decreases surgical morbidity
- Allows for implant dentistry access to a large edentulous or partially edentulous patient population
- Offers an additional innovative procedure for the dental practice
- Designed to increase implant treatment acceptance due to a single day procedure
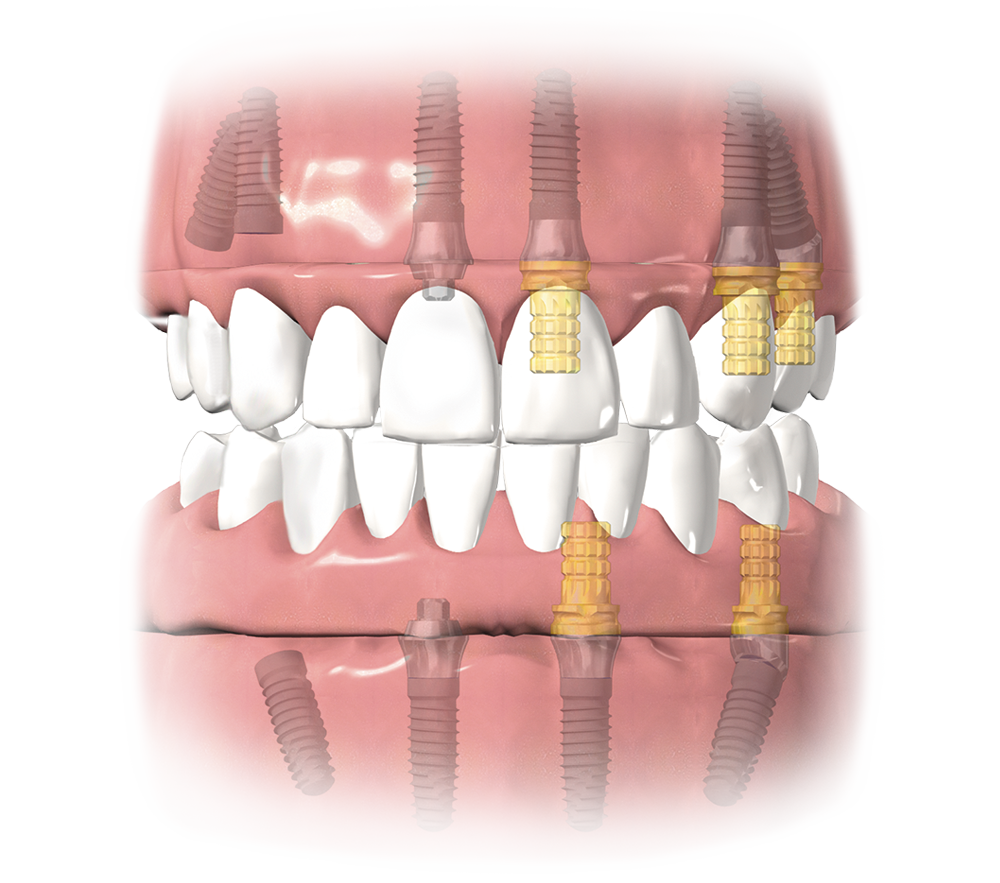

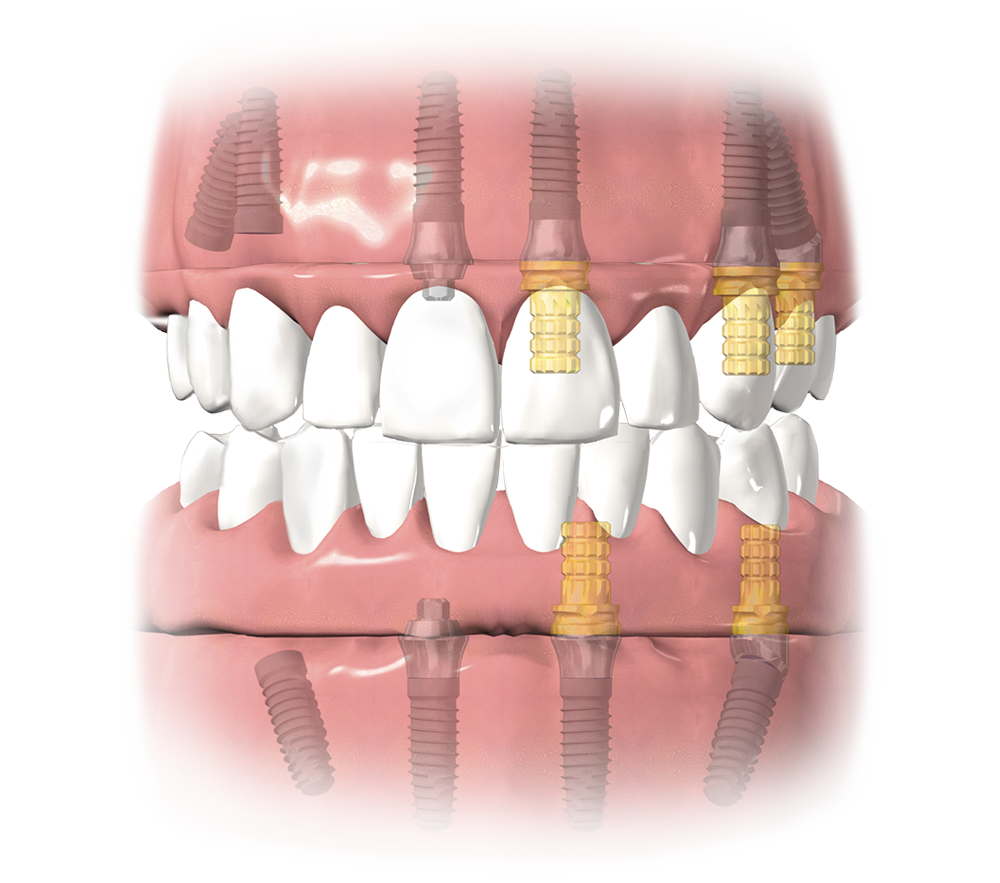
Tapered Implants
Integrated Platform Switching medializes the implant/abutment junction (IAJ) inward, creating a biologic width between connective tissue and the IAJ1- helping to maintain bone levels1

Low Profile Abutments with Prosthetic Components
Good aesthetics are achieved with the gold hue of the Temporary Cylinder through the acrylic resin. Ease of use is provided by machined concavities at the apex of the cylinders to retain the rubber dam.

Bars & Frameworks
A truly passive fit. CAD/CAM precision. No soldering or welding. No waxing and casting. Biocompatible alloy.

DENTAL EDUCATION
ZimVie Institute
Implant skillZTM
Full-Arch Therapy
Resources
Customer Service & Technical Support
At ZimVie Dental, our focus is always on you. Priority attention is the hallmark of our service commitment, and we stand ready to offer you an unprecedented level of service and personalized solutions.